Kiga K. and Cui L.
We humans have developed numerous antimicrobials to combat a variety of bacterial infections. However, the emergence and re-emergence of antimicrobial-resistant bacteria continue to pose a serious threat to global public health and threaten our ability to treat infectious diseases.
In our most recent study in Nature Communications, we have developed a series of RNA-targeted antimicrobials that are capable to kill antimicrobial-resistant bacteria by recognizing corresponding antimicrobial resistance genes. This development is achieved by loading CRISPR-Cas13a programmed to target antimicrobial resistance genes into bacteriophage capsid.
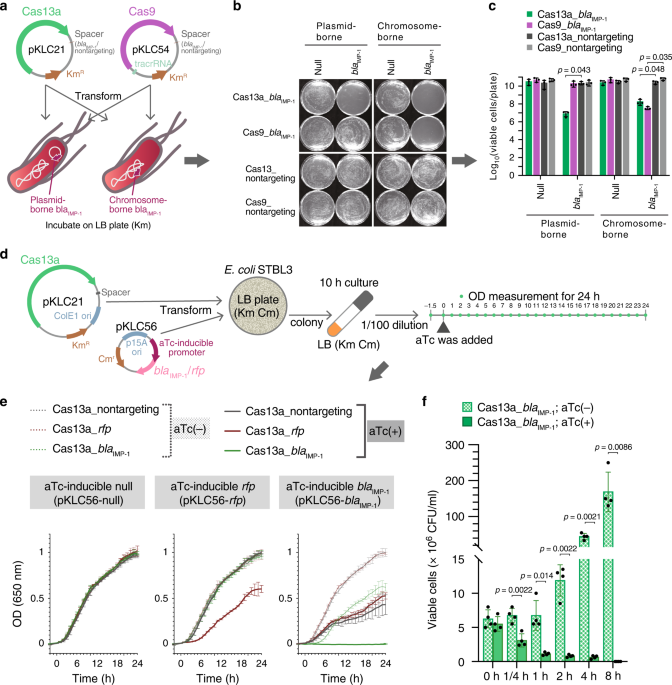
CRISPR-Cas13a is a recently identified CRISPR-Cas system and characterized to have an promiscuous RNA cleavage activity upon sequence-specific binding to a single-stranded RNA molecule. This unique mode of action of the CRISPR-Cas13a prompts us to contemplate its potential as a new antimicrobial. Our team assessed the capabilities of the CRISPR-Cas13a as a potential therapeutic against antimicrobial-resistant bacteria as well as a non-chemical agent for bacterial gene detection.

Having proved a strong bactericidal effect of Cas13a with optimized crRNA spacer sequence, we proceed to generate antibacterial agents by packaging CRISPR-Cas13a genes into bacteriophage. Eventually, a series of CRISPR-Cas13a-based antibacterial nucleocapsids, termed CapsidCas13a(s), capable of sequence-specific killing of carbapenem-resistant Escherichia coli and methicillin-resistant Staphylococcus aureus by recognizing corresponding antimicrobial resistance genes were generated.
We also demonstrated several potential applications that the CapsidCas13a(s) antimicrobials are promising to be developed; i) as antibacterial therapeutic agents targeting any bacterial gene, including antibacterial resistance genes; ii) as a bacterial gene detection system without nucleic acid amplification and optic devices, and iii) as tools to manipulate the bacterial flora by eliminating a certain bacterial population without disrupting irrelevant bacterial populations.
Kiga K., Tan XE., Ibarra-Chávez R., Watanabe S., Aiba Y., Sato’o Y., Li FY., Sasahara T., Cui B., Kawauchi M., Boonsiri T., Thitiananpakorn K., Taki Y., Azam AH., Suzuki M., Penadés JR, Cui L. Development of CRISPR-Cas13a-based antimicrobials capable of sequence-specific killing of target bacteria. Nat Commun, 11, 2934 (2020). https://doi.org/10.1038/s41467-020-16731-6






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in